Structured log capture
Collect consistent event and condition data across the cold chain.
Maintain clear, traceable log records across storage and transit workflows to support review and compliance checks.
Collect consistent event and condition data across the cold chain.
Support recurring operational and compliance review cycles.
Prepare usable evidence for audits and investigations.
This page is aligned to core evidence themes for monitoring, response, and documented review.

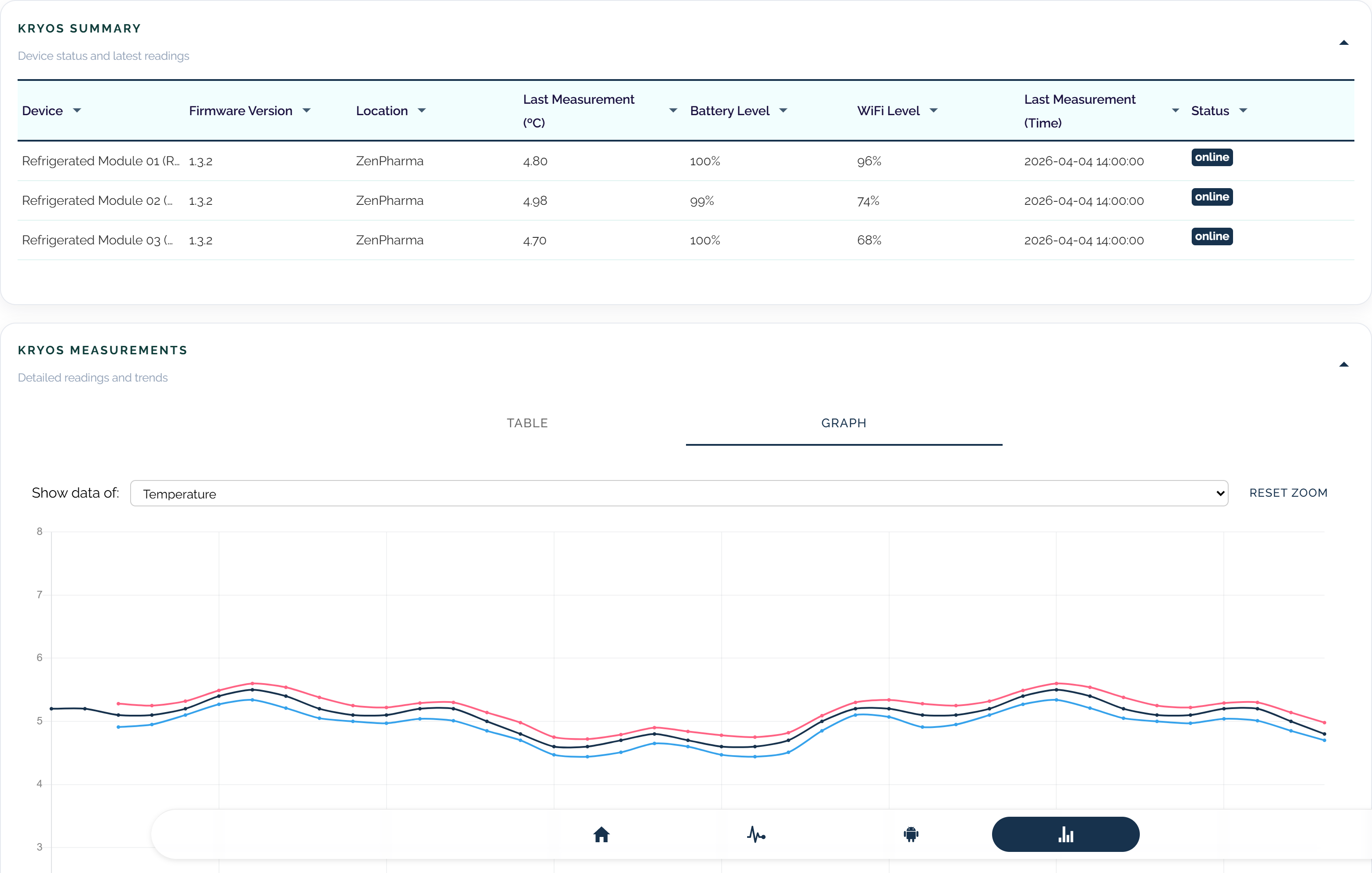
Whether you monitor pharmacy refrigerators, medical storage, or transport conditions, we can help you build a cleaner and more reliable monitoring setup.