Storage condition assurance
Monitor vaccine units continuously with threshold controls.
Maintain vaccine storage oversight with continuous readings, alerts, and structured incident timelines.
Monitor vaccine units continuously with threshold controls.
Capture how teams respond to storage deviations.
Preserve records for regulatory and internal review.
This page is aligned to core evidence themes for monitoring, response, and documented review.

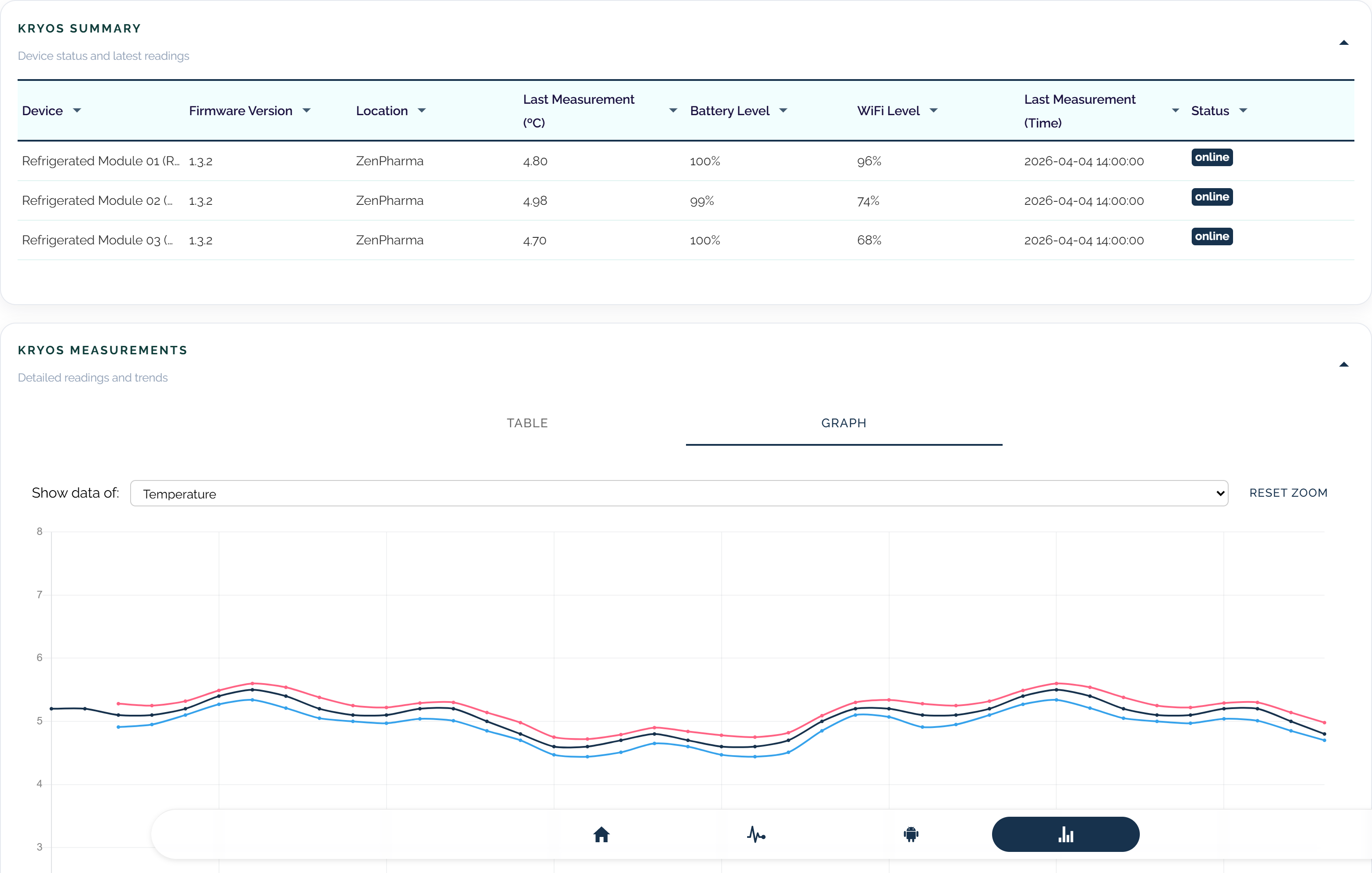
Whether you monitor pharmacy refrigerators, medical storage, or transport conditions, we can help you build a cleaner and more reliable monitoring setup.